|
Electrons are grouped into general regions called shells, and within these into more specific regions called subshells. 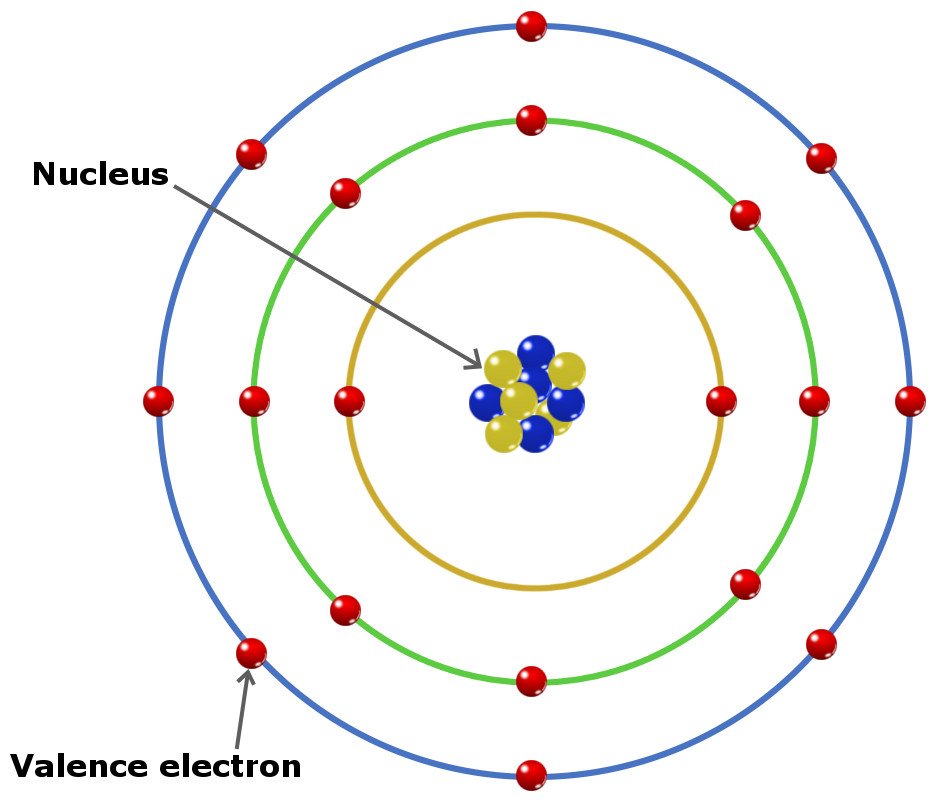
According to this theory, electrons in atoms can only have specific, or quantized, energies. The modern theory of electron behavior is called quantum mechanics. Because different isotopes of an element have different masses, the atomic mass of an element is a weighted average of the masses of all the element’s naturally occurring isotopes. An atomic mass unit is equal to 1/12th of the mass of a single carbon-12 atom. Masses of individual atoms are measured in atomic mass units. The sum of the numbers of protons and neutrons in a nucleus is called the mass number and is used to distinguish isotopes from each other. Two isotopes of hydrogen are deuterium, with a proton and a neutron in its nucleus, and tritium, with a proton and two neutrons in its nucleus. An element may have different numbers of neutrons in the nuclei of its atoms such atoms are referred to as isotopes. This number of protons is the atomic number of the element. The electrons are found in fuzzy clouds around the nucleus.Įach element has a characteristic number of protons in its nucleus. The protons and neutrons are found in the center of the atom, grouped together into a nucleus. Protons, neutrons, and electrons have a specific arrangement in an atom. The neutron is also much larger than an electron but has no electrical charge. The proton has a positive charge and, while small, is much larger than the electron. The electron is a tiny subatomic particle with a negative charge. Atoms themselves are composed of subatomic particles. :max_bytes(150000):strip_icc()/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)
Most elements exist in pure form as individual atoms, but some exist as diatomic molecules. Individual atoms are extremely small, on the order of 10 −10 m across. The modern atomic theory states that the smallest piece of an element is an atom. Each element has a one- or two-letter chemical symbol. They have varying abundances on Earth and in the body. Only about 90 naturally occurring elements are known. To ensure that you understand the material in this chapter, you should review the meanings of the following bold terms and ask yourself how they relate to the topics in the chapter.Īn element is a substance that cannot be broken down into simpler chemical substances.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
